|
7/11/2023 0 Comments Moderna covid vaccine infographic Following the Standard Process, vaccine candidates take 10-15 years before reaching the market. Speeds up the up the Pre- and Clinical Phases part of the Standard Process under public health emergencies without compromising safety. Currently, Pfizer-BioNTech and Moderna SARS-CoV-2 vaccines are both fully approved for ages 18+ in the USA. The continued surveillance of vaccines post-market means that any low-frequency, adverse events not apparent in clinical trials can still be caught and the FDA can halt administration of the vaccine while the new data are assessed. In general, the vaccines will be monitored for safety through multiple surveillance systems under the FDA and CDC such as the Vaccine Adverse Event Reporter System (VAERS) and the FDA Biologicals Effectiveness and Safety (BEST) program. In some cases, the FDA may have a manufacturer specifically conduct post-marketing studies (a Phase IV trial). After careful review, the FDA will decide on approval.Īpproved vaccines will be manufactured, marketed, and administered to the population(s) the vaccine was intended for, though the FDA will continue to monitor the manufacturing and safety of the vaccines. The VRBPAC provides an additional review of the vaccine that the FDA will consider in deciding to grant approval. Unlike the FDA review panel, the VRBPAC committee is made of independent, scientific, or public health experts. In some instances, the FDA will also receive input from its Vaccines and Related Biological Products Advisory Committee (VRBPAC). The team of FDA reviewers consists of a broad range of specialists from physicians and toxicologists to manufacturing and facility inspectors. The FDA will look over the vaccine data and assess the risks and benefits for the vaccine in its intended target population. Vaccine candidates that were successful in meeting their goals during the clinical trials will be submitted under a Biological License Application (BLA) to the FDA for review. 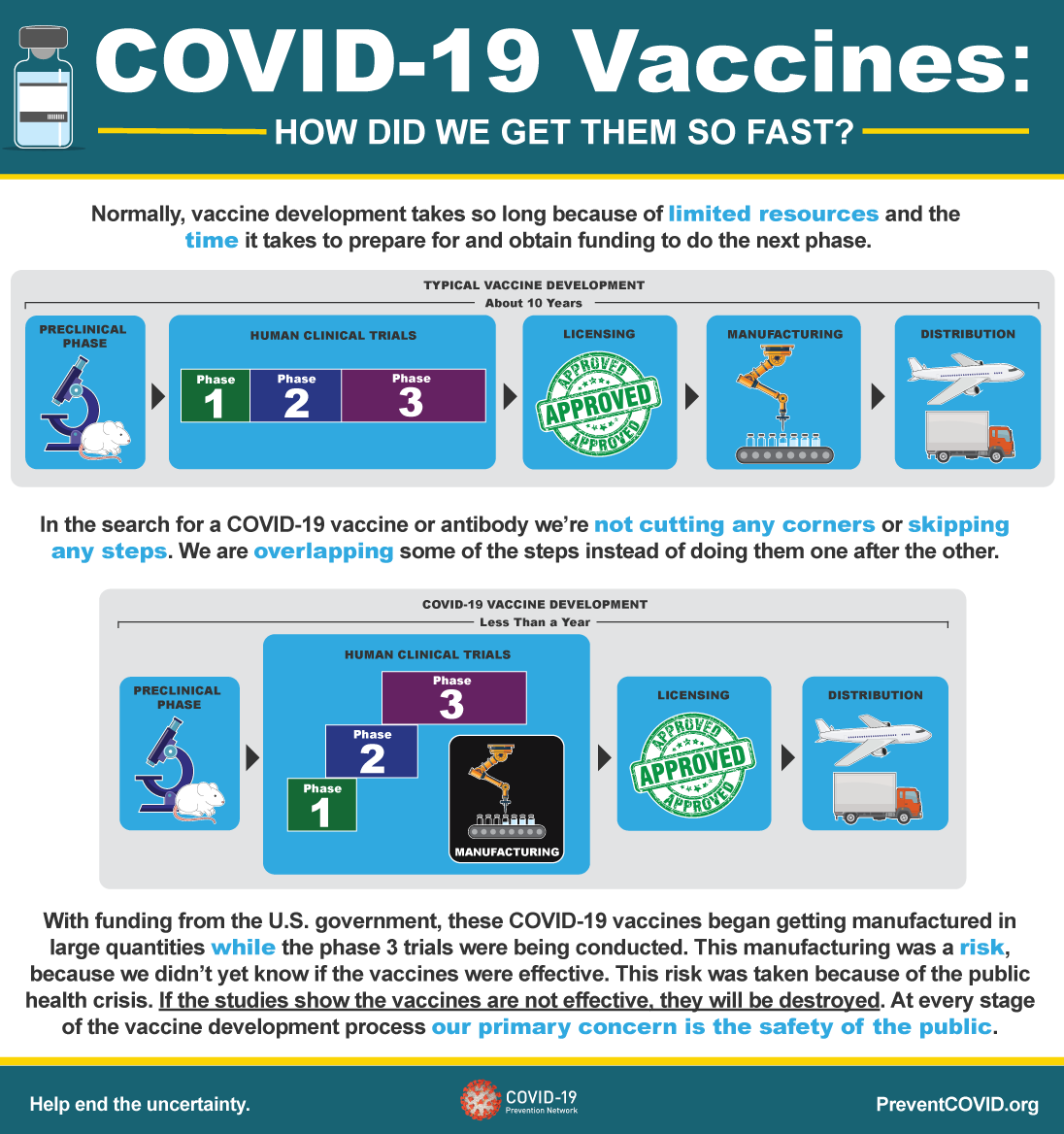
Additionally, phases may be repeated as necessary if the first round did not meet its efficacy goals. An important part to note in the Standard Process is that researchers analyze the data after each phase has ended before progressing to the next phase. 
The clinical phases may be single- or double-blinded to help prevent researcher bias.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
